Otsuka Pharmaceutical Co., Ltd.
New Drug Application Approval of TAKECAB® for the Treatment of Acid-related Diseases in Japan
- Takeda received approval of its New Drug Application for TAKECAB® for the treatment of acid-related diseases in Japan.
- Takeda and Otsuka executed an agreement at the end of March 2014 to co-promote TAKECAB® in Japan. Under the agreement, the two companies will conduct informational activities for healthcare professionals with the aim of addressing unmet clinical needs in the treatment of acid-related diseases.
- At this time, Takeda will receive a milestone payment for the NDA approval. Otsuka will receive co-promotion fee based on sales amounts (in accordance with certain conditions specified in the agreement) from Takeda.
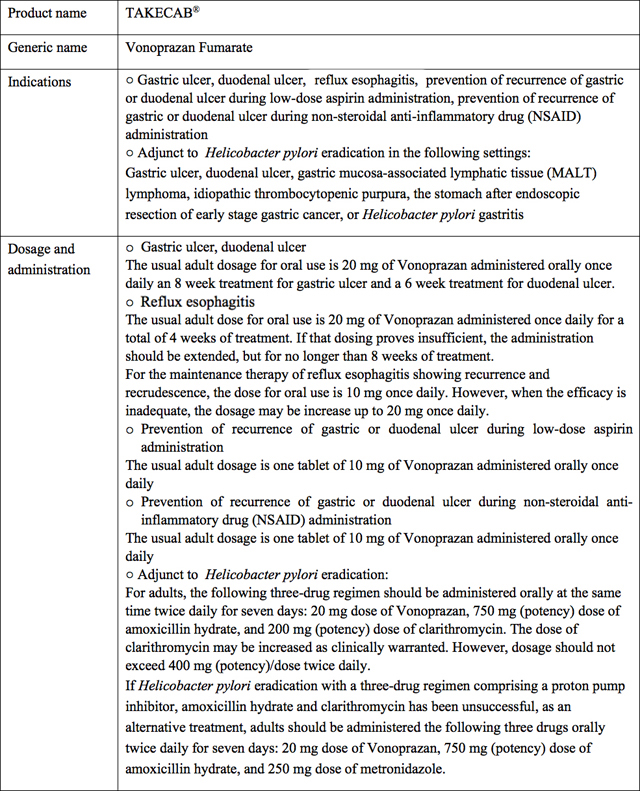
Takeda Pharmaceutical Company Limited (Head office: Chuo-ku, Osaka; President and COO: Christophe Weber; "Takeda") and Otsuka Pharmaceutical Company, Limited (Head office: Chiyoda-ku, Tokyo; President and Representative Director: Taro Iwamoto; "Otsuka") announce today that the Japanese Ministry of Health, Labour and Welfare has approved the New Drug Application submitted by Takeda ("NDA") of TAKECAB® (generic name: Vonoprazan fumarate, hereafter "TAKECAB"), a drug for treating acid-related diseases.
TAKECAB, discovered by Takeda, is a new medicine for treating acid-related diseases with a novel mechanism of action called potassium-competitive acid blockers (P-CABs) which competitively inhibits the binding of potassium ions to H+,K+-ATPase (also known as the proton pump) in the final step of gastric acid secretion in gastric parietal cells. TAKECAB provides a strong and sustained acid secretion inhibitory effect.
The approval granted is based on the results of multiple Phase III clinical trials for TAKECAB in Japan. Takeda has conducted trials for indications including gastric ulcer, duodenal ulcer, reflux esophagitis and H. pylori eradication. In these trials, TAKECAB demonstrated efficacy and has a favorable profile for safety and tolerability.
Takeda and Otsuka executed an agreement at the end of March 2014 to co-promote TAKECAB in Japan. Under the agreement, Takeda will manufacture and market TAKECAB, and Takeda and Otsuka will jointly conduct informational activities for healthcare professionals. Through this partnership, the two companies aim to address issues related to the treatment of acid-related diseases and address unmet clinical needs in the gastrointestinal field.
References
About the co-promotion agreement
The details of this agreement are as below:
- Takeda will receive from Otsuka an up-front payment of 20 billion yen and a milestone payment upon regulatory approval.
- Otsuka will receive from Takeda co-promotion fee based on the sales amount (based on conditions specified in the contract).
- Territory: Japan
Further details are not disclosed.





